
- Biotechnology
- CRISPR Gene-Editing Market
CRISPR Gene-Editing Market Size, Share, and Growth Forecast 2026 - 2033
CRISPR Gene-Editing Market by Product (Kits & Enzymes, Libraries, Others), Service (gRNA Design and Vector Construction, Cell Line Engineering, Screening Services, Other), Application, and Regional Analysis, 2026 - 2033
CRISPR Gene-Editing Market Size and Trend Analysis
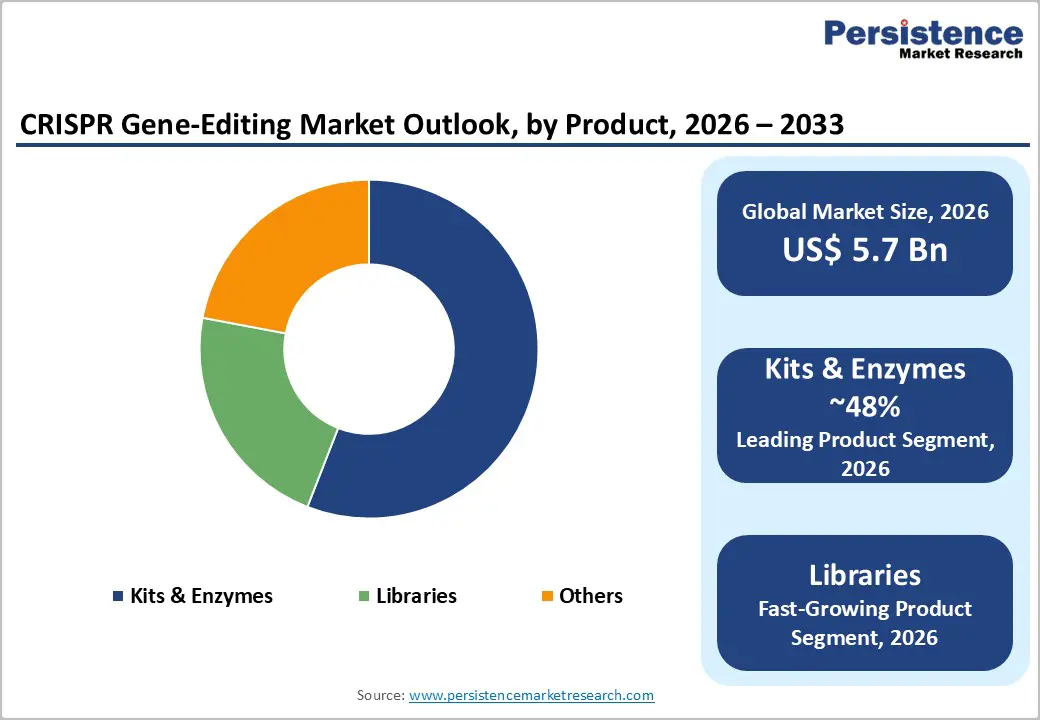
The global CRISPR Gene-Editing Market size is expected to be valued at US$ 5.7 billion in 2026 and projected to reach US$ 12.4 billion by 2033, growing at a CAGR of 11.8% between 2026 and 2033.
The rising demand for CRISPR-based gene editing to treat genetic illnesses, the development of vaccines and novel diagnostic tools, and the modification of crops for higher yield and quality drive the need for gene-editing techniques. CRISPR gene-editing enables scientists to change an organism's DNA by adding, removing, or altering genetic material at specific locations in the genome. The well-known gene-editing approach is called CRISPR-Cas9, which is short for clustered regularly interspaced short palindromic repeats and CRISPR-associated protein 9. It is faster, cheaper, more accurate, and more efficient than other genome editing methods. As per the Britannica article published in May 2025, CRISPR-Cas9 has been used to edit early embryos and to modify the genomes of crops, animals, and bacteriophages to combat antibiotic-resistant bacteria. It has corrected genetic defects in mice and removed HIV from infected cells. These developments highlight the significant growth potential of the market.
Key Industry Highlights:
- The market is driven by rising demand for CRISPR-based gene editing to treat genetic illnesses, the development of vaccines and novel diagnostic tools, and the modification of crops for improved yield and quality.
- The kits & enzymes segment is projected to dominate due to their wide-ranging applications in genome modification, gene knockout, and validation studies.
- The libraries segment is poised for rapid growth as they are used for high-throughput screening to identify genes involved in specific biological processes or to study how cells respond to various conditions.
- North America dominated the global market due to the presence of well-established R&D centers and increased funding for innovation and clinical trials.
- CRISPR gene-editing enables the precise targeting of genes that cause or contribute to the progression of genetic diseases.
- CRISPR significantly expands the scope and impact of regenerative and precision medicine by creating next-generation cell therapies.
| Key Insights | Details |
|---|---|
|
CRISPR Gene-Editing Market Size (2026E) |
US$ 5.7 billion |
|
Market Value Forecast (2033F) |
US$ 12.4 billion |
|
Projected Growth CAGR (2026-2033) |
11.8% |
|
Historical Market Growth (2020-2025) |
8.5% |

Market Dynamics
Drivers - Rapid clinical translation of CRISPR-based therapeutics
A key driver for the CRISPR gene-editing market is the rapid transition of genome-editing technologies from the research setting to approved therapies. In December 2023, the FDA approved CASGEVY (exa-cel), the first CRISPR/Cas9-based gene therapy, for sickle cell disease in patients 12 years and older with recurrent vaso-occlusive crises, marking a historic inflection point for the field. Clinical data show that treated patients experienced a near-elimination of severe vaso?occlusive events and sustained increases in fetal hemoglobin levels, demonstrating the curative potential of one-time CRISPR interventions. In addition, global pipelines now include approximately 250 gene-editing clinical trials with more than 150 active studies across oncology, hematology, ophthalmology, infectious diseases, and rare disorders, significantly expanding the addressable market for CRISPR tools, reagents, and enabling services. As more programs move into Phase II and Phase III, demand rises for high-quality kits & enzymes, sophisticated libraries, and specialized services that support translational research and clinical manufacturing.
Rising demand for CRISPR gene editing in treating illnesses
CRISPR technology is transforming medicine through precise genome editing, enabling innovations in genetic disease correction, oncology, regenerative medicine, and infectious disease treatment. The December 2024 study published in Frontiers stated that the advances in CRISPR gene-editing, such as high-fidelity Cas9, base/prime editors, and Cas12/Cas13, have improved accuracy and efficiency. Emerging epigenome editors allow gene regulation without changing DNA, offering safer and reversible treatment options. The FDA approval of the treatment of sickle cell disease using Casgevy highlights its high clinical potential. Recent breakthroughs highlight CRISPR’s rapidly growing role across diverse fields, positioning it as a major growth driver in the gene-editing market.
CRISPR gene-editing has been used to repair genetic mutations and combat antibiotic-resistant bacteria. In biomedical research and therapeutics, CRISPR interventions are revolutionary. For instance, in May 2025, a U.S. infant with CPS1 deficiency became the first human to receive a personalized gene-editing treatment, marking a significant milestone in rare disease therapeutics. This achievement highlights CRISPR's growing potential in personalized medicine and its capacity to treat rare conditions.
Restraints - Ethical concerns pertaining to CRISPR gene-editing to hamper its widespread adoption
Ethical concerns surrounding gene editing remain a major restraint for the CRISPR gene-editing market. The CRISPR-Cas9 system enables precise genetic modifications and plays an important role in Human Germline Engineering (HGE). However, germline editing involves modifying embryos or reproductive cells, meaning genetic changes can be inherited by future generations. While somatic cell editing is widely accepted for therapeutic purposes, germline modifications raise significant moral and ethical concerns.
One key issue is the possibility of non-therapeutic applications, such as altering physical traits like eye color or intelligence, which could lead to the concept of “designer babies.” Additionally, ethical debates arise because genome-edited children cannot provide consent for genetic changes that may permanently affect their lives and future generations. Regulatory differences across countries further complicate adoption. According to studies referenced by the National Library of Medicine, several countries, including China, the United States, and the United Kingdom, permit limited embryo editing for research purposes, while others, such as Canada, Sweden, and Switzerland, have banned human embryo gene-editing trials. Beyond medical ethics, CRISPR also raises ecological concerns. Technologies like gene drives, designed to eliminate disease-carrying insects such as the Mosquito, could unintentionally disrupt ecosystems, underscoring the need for careful governance and responsible innovation.
Opportunities - R&D Opportunities to Develop Medicines, Vaccines, and Genetically Modified Crops
CRISPR gene-editing allows the precise targeting of specific genes that cause or influence the progression of genetic diseases. As the field evolves and the gene-disease relationships become clearer, gene-editing will help in amending faulty genetic sequences, thereby addressing the root causes of all types of rare and common disorders. Cell therapies, which are already successful in treating diseases such as cancer can be further modified at the genetic level to enhance their safety, efficacy, and accessibility. It is being explored in research and clinical trials for single-gene disorders such as cystic fibrosis, hemophilia, and sickle cell disease. It also holds promise for the treatment and prevention of cancer, heart disease, mental illness, and HIV infection.
CRISPR has the capability to expand the scope and impact of regenerative and precision medicine by creating next-generation cell therapies. Scientists at the University of Bayreuth successfully developed the first CRISPR-Cas9-modified spider capable of producing red, fluorescent silk, an innovation published in Angewandte Chemie International Edition (May 2025). This success illustrates CRISPR’s versatility and its potential for use in novel biomaterials and synthetic biology. Such advancements are driving increased investments in research across therapeutic, agricultural, and industrial biotechnology sectors.
Category-wise Analysis
Product Insights
By product, the kits & enzymes segment is projected to dominate the CRISPR gene-editing market with an estimated 48% share in 2026, owing to their extensive use in genome modification, gene knockout, and validation studies. According to a 2023 study published by the National Library of Medicine, CRISPR kits include essential reagents such as Cas9 enzymes, guide RNAs (gRNAs), and supporting components that provide a pre-optimized system for efficient gene editing. These enzymes work with gRNAs to accurately identify and modify targeted DNA sequences, allowing researchers to precisely edit genes for various applications. Research published in May 2024 in BioMed Central highlighted that CRISPR-associated enzymes such as Cas9 are widely used to identify and correct genetic mutations involved in tumor development and metastasis, strengthening their importance in cancer research and therapeutic innovation.
The libraries segment is expected to be the fastest-growing category during the forecast period. In CRISPR research, libraries consist of large collections of guide RNAs designed to target multiple genes across a genome. These libraries support high-throughput screening to identify gene functions and cellular responses. Two major types include genomic libraries and cDNA libraries, which help researchers study genome structures, gene expression, disease-related mutations, and protein production for biomedical research.
Application Insights
Based on application, the drug discovery & development segment dominates the CRISPR gene-editing market, accounting for approximately 65% of revenue share in 2025. CRISPR technology plays a crucial role in advancing precision medicine by enabling researchers to identify and modify genes associated with diseases. It supports personalized cancer therapies by targeting patient-specific genetic drivers responsible for tumor growth and treatment resistance. The CRISPR-Cas9 system has shown strong potential in treating monogenic disorders such as Sickle Cell Anemia and Beta Thalassemia, while ongoing research is exploring its application for conditions including Huntington’s Disease and HIV/AIDS.
CRISPR is also transforming oncology research through targeted immunotherapy and cancer gene mapping. For instance, research conducted by PACT Pharma in collaboration with the University of California demonstrated the potential of CRISPR-edited T cells to attack cancer cells, where several participants with treatment-resistant cancers experienced disease stabilization. The agriculture segment is projected to be the fastest-growing application area. CRISPR enables precise modifications in plant DNA to develop crops with improved traits such as drought tolerance, disease resistance, and enhanced nutrition. For example, the crop Camelina sativa has been developed using CRISPR technology to produce higher levels of omega-3 oil.
Regional Insights
North America CRISPR Gene-Editing Market Trends and Insights
North America is expected to dominate the global CRISPR gene-editing market in 2025, accounting for nearly 44% of total revenue. The region’s strong biotechnology ecosystem, well-established research infrastructure, and increasing funding for gene-editing innovation and clinical trials are key factors driving market growth. The United States hosts numerous biotechnology and pharmaceutical companies as well as Contract Research Organizations (CROs), including Applied StemCell, Cellecta, Integrated DNA Technologies, and Horizon Discovery, which play an important role in supporting research and commercialization of CRISPR technologies. Additionally, a supportive regulatory environment that encourages innovation and facilitates faster approval of novel therapies further accelerates market expansion.
The U.S. is expected to account for the largest share of the North American CRISPR market in 2025. Strong research initiatives and favorable policies are contributing to the rapid adoption of gene-editing technologies. In the U.S., gene-edited crops developed through CRISPR are regulated based on the final product rather than the editing method. Policies such as the SECURE Rule and investments in the Precision Medicine Initiative are also promoting personalized healthcare and genomic research.
Europe CRISPR Gene-Editing Market Trends and Insights
Europe represents a scientifically advanced and steadily expanding region in the CRISPR gene-editing market, supported by strong academic research, coordinated funding programs, and a structured regulatory environment. Institutions across countries such as Germany, UK, France, and Spain play a key role in advancing genome-editing research and clinical applications. The European Medicines Agency (EMA) has introduced accelerated regulatory pathways such as PRIME designation and orphan drug status to support faster development of gene therapy and genome-editing treatments for rare and life-threatening diseases.
At the same time, the European Union maintains strict regulations for genetically modified organisms (GMOs), and ongoing policy discussions continue regarding how gene-edited crops should be regulated compared with transgenic GMOs. This careful regulatory approach ensures safety and environmental protection while allowing innovation in medical biotechnology.
Across Europe, numerous academic collaborations and biotechnology startups are focusing on delivery technologies, base editing, and ethical, legal, and social implications of human genome editing. Although regulatory complexity may extend development timelines compared with North America, Europe’s strengths in translational science, bioethics, and advanced biomanufacturing position the region as an important contributor to global CRISPR innovation.
Asia Pacific CRISPR Gene-Editing Market Trends and Insights
The Asia Pacific CRISPR gene-editing market is projected to grow at the fastest pace during the forecast period, driven by rising investments in biotechnology research, expanding clinical trials, and increasing adoption of advanced genetic technologies. Countries such as China and India are playing a major role in accelerating regional growth through strong government support and expanding biotechnology ecosystems. The growing prevalence of chronic and genetic diseases, along with increasing positive clinical trial outcomes, is attracting substantial investments in gene-editing research.
Several biotechnology companies, including OliX Pharmaceuticals, Suzhou Ribo Life Science, Gennova Biopharmaceuticals, Nuevocor, and Abion, are actively developing gene therapy and RNA-based therapeutic solutions in the region. In addition, Rznomics from South Korea is developing multiple pipeline drugs using trans-splicing ribozyme technology to treat cancer, neurodegenerative diseases, and genetic eye disorders. One of its candidates, RZ-001, has entered Phase I/IIa trials for hepatocellular carcinoma and glioblastoma.
China’s gene-editing industry is expanding rapidly due to strong research investments, supportive policies, and the presence of large pharmaceutical companies, making the country a key hub for CRISPR innovation.

Competitive Landscape
The global CRISPR gene-editing market is moderately fragmented, with several key players driving growth through product innovation, strategic partnerships, and acquisitions. Continued investments in R&D to develop new products are expected to strengthen their market position over the forecast period.
Key Developments:
- In January 2026, Aurora Therapeutics, co-founded by Nobel Prize–winning scientist Jennifer Doudna, launched a startup aimed at developing customized gene-editing therapies. The company plans to leverage advanced gene-editing technologies and a new regulatory pathway introduced by the U.S. Food and Drug Administration to accelerate the commercialization of treatments for rare diseases.
- In May 2025, NKure Therapeutics and CRISPR Therapeutics announced a partnership to co-develop and co-commercialize CTX112, a next-generation off-the-shelf CAR-T therapy for oncology indications in India.
- In January 2025, ERS Genomics launched its Express License platform as a strategic initiative to support small research enterprises and startups, offering affordable and rapid access to the world’s largest foundational and essential CRISPR/Cas9 patent portfolio.
Companies Covered in CRISPR Gene-Editing Market
- Revvity, Inc.
- Danaher
- GenScript
- Merck KGaA
- Thermo Fisher Scientific, Inc.
- Tocris Bioscience
- OriGene Technologies, Inc.
- Bio-Rad Laboratories
- Bio-Techne
- New England Biolabs, Inc.
Frequently Asked Questions
The global CRISPR Gene-editing market is projected to be valued at US$ 5.7 billion in 2026.
Rising demand for genetic disease therapies, advanced diagnostics, vaccine development, and crop improvement drives CRISPR gene-editing market growth.
North America leads the CRISPR gene-editing market due to strong research funding, advanced biotech ecosystem, and early regulatory approvals.
R&D efforts are underway to allow the precise targeting of specific genes that cause or influence the progression of genetic diseases.
Major players in the CRISPR gene-editing industry include Revvity, Inc., Danaher, GenScript, Merck KGaA, and Thermo Fisher Scientific, Inc.







