- Executive Summary
- Global Liver Cirrhosis Treatment Market Snapshot, 2026 and 2033
- Market Opportunity Assessment, 2026 – 2033, US$ Bn
- Key Market Trends
- Future Market Projections
- Premium Market Insights
- Industry Developments and Key Market Events
- PMR Analysis and Recommendations
- Market Overview
- Market Scope and Definition
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Challenges
- Key Trends
- COVID-19 Impact Analysis
- Forecast Factors – Relevance and Impact
- Value Added Insights
- Value Chain Analysis
- Key Market Players
- Regulatory Landscape
- PESTLE Analysis
- Porter’s Five Force Analysis
- Consumer Behavior Analysis
- Price Trend Analysis, 2020-2025
- Key Factors Impacting Product Prices
- Pricing Analysis, By Treatment Type
- Regional Prices and Product Preferences
- Global Liver Cirrhosis Treatment Market Outlook
- Market Size (US$ Bn) Analysis and Forecast
- Historical Market Size (US$ Bn) Analysis, 2020-2025
- Market Size (US$ Bn) Analysis and Forecast, 2026–2033
- Global Liver Cirrhosis Treatment Market Outlook: Treatment Type
- Historical Market Size (US$ Bn) Analysis, By Treatment Type, 2020-2025
- Market Size (US$ Bn) Analysis and Forecast, By Treatment Type, 2026–2033
- Antibiotics
- Immunosuppressants
- Others
- Market Attractiveness Analysis: Treatment Type
- Global Liver Cirrhosis Treatment Market Outlook: End-user
- Historical Market Size (US$ Bn) Analysis, By End-user, 2020-2025
- Market Size (US$ Bn) Analysis and Forecast, By End-user, 2026–2033
- Hospitals & Clinics
- Research and Academic Institutes
- Others
- Market Attractiveness Analysis: End-user
- Market Size (US$ Bn) Analysis and Forecast
- Global Liver Cirrhosis Treatment Market Outlook: Region
- Historical Market Size (US$ Bn) Analysis, By Region, 2020-2025
- Market Size (US$ Bn) Analysis and Forecast, By Region, 2026–2033
- North America
- Latin America
- Europe
- East Asia
- South Asia and Oceania
- Middle East & Africa
- Market Attractiveness Analysis: Region
- North America Liver Cirrhosis Treatment Market Outlook
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- By Country
- By Treatment Type
- By End-user
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026–2033
- U.S.
- Canada
- Market Size (US$ Bn) Analysis and Forecast, By Treatment Type, 2026–2033
- Antibiotics
- Immunosuppressants
- Others
- Market Size (US$ Bn) Analysis and Forecast, By End-user, 2026–2033
- Hospitals & Clinics
- Research and Academic Institutes
- Others
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- Europe Liver Cirrhosis Treatment Market Outlook
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- By Country
- By Treatment Type
- By End-user
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026–2033
- Germany
- France
- U.K.
- Italy
- Spain
- Russia
- Rest of Europe
- Market Size (US$ Bn) Analysis and Forecast, By Treatment Type, 2026–2033
- Antibiotics
- Immunosuppressants
- Others
- Market Size (US$ Bn) Analysis and Forecast, By End-user, 2026–2033
- Hospitals & Clinics
- Research and Academic Institutes
- Others
- Market Attractiveness Analysis
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- East Asia Liver Cirrhosis Treatment Market Outlook
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- By Country
- By Treatment Type
- By End-user
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026–2033
- China
- Japan
- South Korea
- Market Size (US$ Bn) Analysis and Forecast, By Treatment Type, 2026–2033
- Antibiotics
- Immunosuppressants
- Others
- Market Size (US$ Bn) Analysis and Forecast, By End-user, 2026–2033
- Hospitals & Clinics
- Research and Academic Institutes
- Others
- Market Attractiveness Analysis
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- South Asia & Oceania Liver Cirrhosis Treatment Market Outlook
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- By Country
- By Treatment Type
- By End-user
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026–2033
- India
- Indonesia
- Thailand
- Singapore
- ANZ
- Rest of South Asia & Oceania
- Market Size (US$ Bn) Analysis and Forecast, By Treatment Type, 2026–2033
- Antibiotics
- Immunosuppressants
- Others
- Market Size (US$ Bn) Analysis and Forecast, By End-user, 2026–2033
- Hospitals & Clinics
- Research and Academic Institutes
- Others
- Market Attractiveness Analysis
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- Latin America Liver Cirrhosis Treatment Market Outlook
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- By Country
- By Treatment Type
- By End-user
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026–2033
- Brazil
- Mexico
- Rest of Latin America
- Market Size (US$ Bn) Analysis and Forecast, By Treatment Type, 2026–2033
- Antibiotics
- Immunosuppressants
- Others
- Market Size (US$ Bn) Analysis and Forecast, By End-user, 2026–2033
- Hospitals & Clinics
- Research and Academic Institutes
- Others
- Market Attractiveness Analysis
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- Middle East & Africa Liver Cirrhosis Treatment Market Outlook
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- By Country
- By Treatment Type
- By End-user
- Market Size (US$ Bn) Analysis and Forecast, By Country, 2026–2033
- GCC Countries
- Egypt
- South Africa
- Northern Africa
- Rest of Middle East & Africa
- Market Size (US$ Bn) Analysis and Forecast, By Treatment Type, 2026–2033
- Antibiotics
- Immunosuppressants
- Others
- Market Size (US$ Bn) Analysis and Forecast, By End-user, 2026–2033
- Hospitals & Clinics
- Research and Academic Institutes
- Others
- Market Attractiveness Analysis
- Historical Market Size (US$ Bn) Analysis, By Market, 2020-2025
- Competition Landscape
- Market Share Analysis, 2025
- Market Structure
- Competition Intensity Mapping By Market
- Competition Dashboard
- Company Profiles (Details – Overview, Financials, Strategy, Recent Developments)
- Gilead Sciences
- Overview
- Segments and Treatment Type
- Key Financials
- Market Developments
- Market Strategy
- AbbVie
- Bristol-Myers Squibb
- Merck & Co.
- Novartis
- Roche
- Amgen
- Eisai
- Hepion Pharmaceuticals
- Gilead Sciences
- Appendix
- Research Methodology
- Research Assumptions
- Acronyms and Abbreviations
- Pharmaceuticals
- Liver Cirrhosis Treatment Market
Liver Cirrhosis Treatment Market Size, Share, and Growth Forecast, 2026 - 2033
Liver Cirrhosis Treatment Market by Treatment (Antibiotics, Immunosuppressants, Others), End-user (Hospitals & Clinics, Research and Academic Institutes, Others), and Regional Analysis for 2026 – 2033
Liver Cirrhosis Treatment Market Size and Trends Analysis
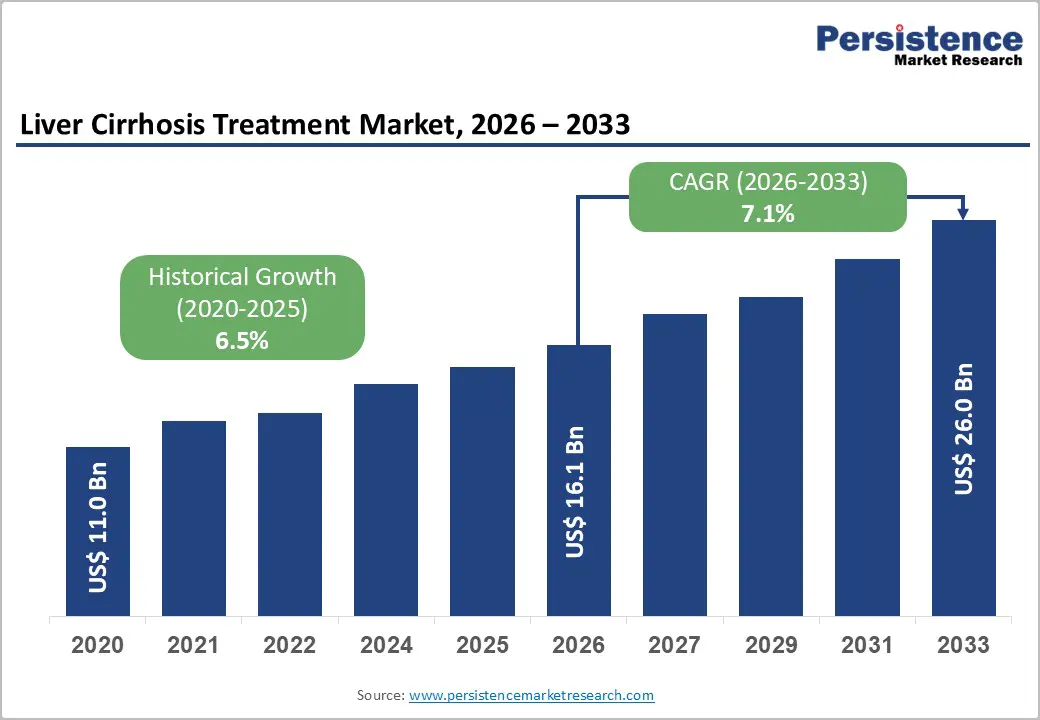
The global liver cirrhosis treatment market size is likely to be valued at US$16.1 billion in 2026, and is expected to reach US$26.0 billion by 2033, growing at a CAGR of 7.1% during the forecast period from 2026 to 2033, driven by the expanding treatment landscape for viral hepatitis, the rising global prevalence of non-alcoholic steatohepatitis (NASH), and the growing number of liver transplant programs aimed at managing advanced liver disease. Ongoing development of antifibrotic drugs is creating new opportunities to slow fibrosis progression and improve patient outcomes.
Rising demand for effective management of cirrhosis complications such as ascites, variceal bleeding, and hepatic encephalopathy, along with the growing use of combination treatment regimens, is supporting market growth. Increasing adoption of antibiotics and immunosuppressants in hospitals and clinics, particularly for decompensated cirrhosis and post-transplant care, further strengthens demand. Additionally, advances in rifaximin formulations and carvedilol-based prophylaxis are helping improve survival, reduce hospitalizations, and enhance patient quality of life.
Key Industry Highlights:
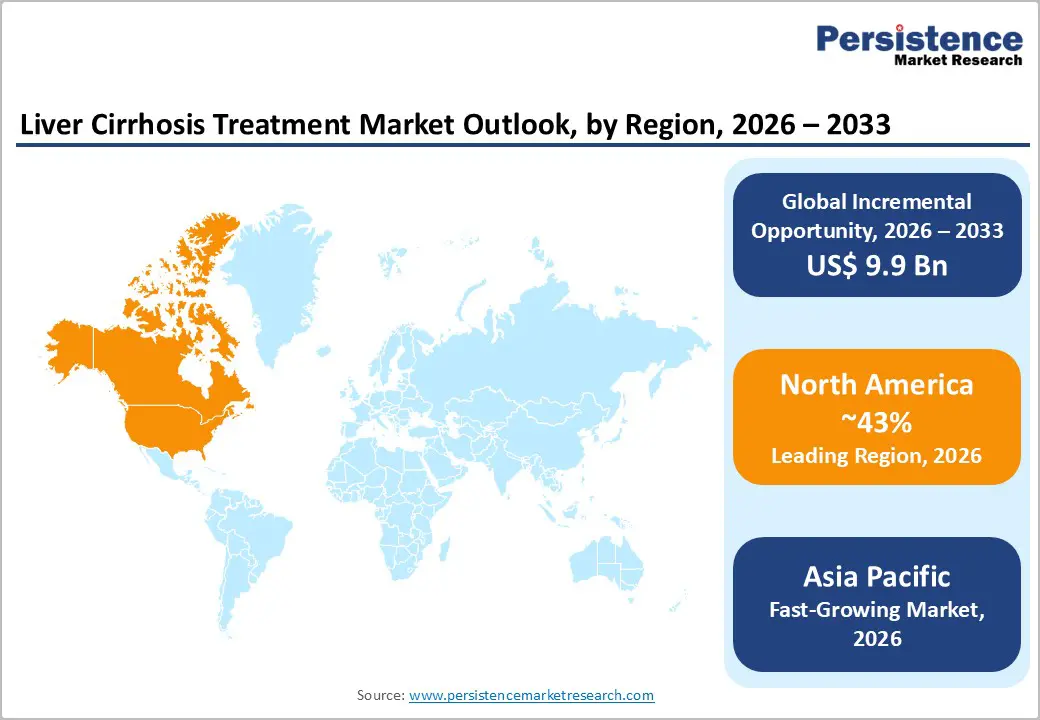
- Leading Region: North America, anticipated to account for a 43% market share in 2026, driven by high NASH and alcohol-related cirrhosis burden, advanced transplant centers, and strong reimbursement in the U.S.
- Fastest-growing Region: Asia Pacific, fueled by massive hepatitis B/C prevalence, rising alcohol consumption, and rapid hospital infrastructure expansion in India and China.
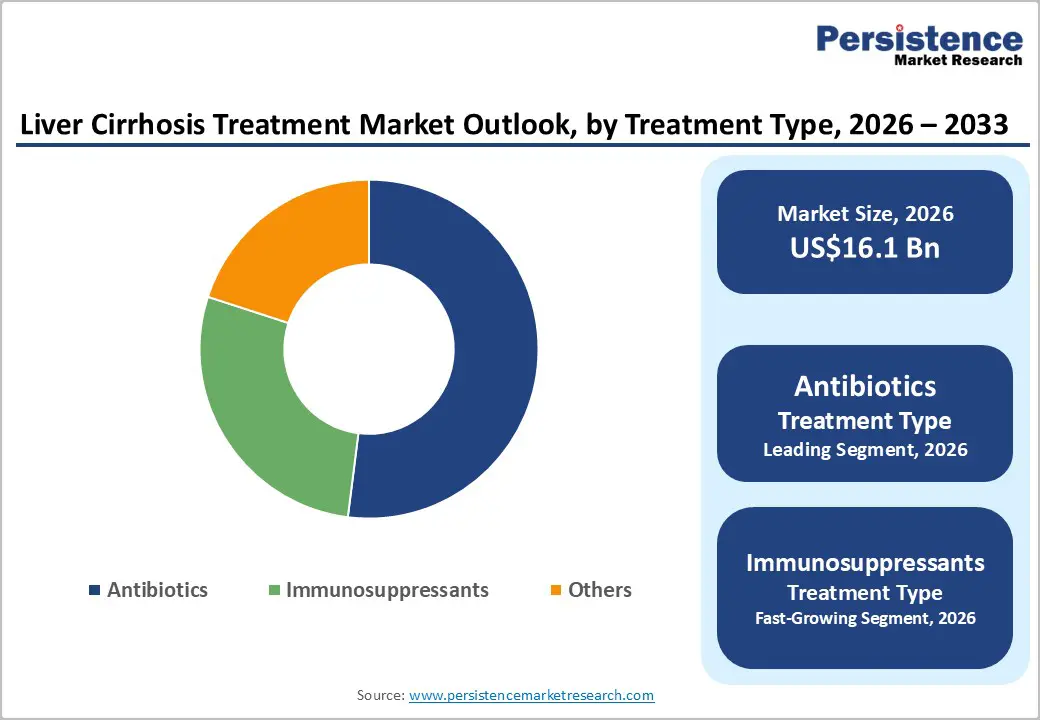
- Dominant Treatment Type: Antibiotics, to hold approximately 52% of the market share, as they remain the cornerstone for spontaneous bacterial peritonitis and hepatic encephalopathy.
- Leading End-user: Hospitals & clinics, to contribute nearly 65% of the market revenue, due to centralized management of decompensated cases.
| Key Insights | Details |
|---|---|
| Liver Cirrhosis Treatment Market Size (2026E) | US$16.1 Bn |
| Market Value Forecast (2033F) | US$26.0 Bn |
| Projected Growth CAGR (2026-2033) | 7.1% |
| Historical Market Growth (2020-2025) | 6.5% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Growth Analysis – Escalating NASH and Alcohol-Related Cirrhosis Burden
Non-alcoholic steatohepatitis (NASH) and alcohol-related cirrhosis are increasingly contributing to the global liver disease burden due to lifestyle, dietary, and alcohol consumption patterns. NASH, a progressive form of non-alcoholic fatty liver disease (NAFLD), arises from excessive fat accumulation in the liver, often associated with obesity, insulin resistance, and metabolic syndrome. Its prevalence is rising sharply, particularly in developed and rapidly urbanizing countries, as sedentary lifestyles and calorie-dense diets become more common. NASH can progress silently to fibrosis, cirrhosis, and hepatocellular carcinoma, making early detection challenging.
Alcohol-related cirrhosis remains a major cause of liver morbidity and mortality worldwide, fueled by chronic excessive alcohol intake. Socioeconomic stressors, cultural norms, and increasing alcohol availability exacerbate the risk. Both NASH and alcohol-induced liver injury place a substantial strain on healthcare systems due to hospitalization, liver transplantation needs, and long-term disease management. The dual escalation of NASH and alcohol-related cirrhosis underscores the urgent need for public health interventions, including lifestyle modification programs, early screening strategies, and awareness campaigns targeting high-risk populations.
Expanding Transplant Waitlist and Bridge Therapies
The liver cirrhosis treatment landscape is increasingly shaped by the growing transplant waitlist, reflecting both rising disease prevalence and limited organ availability. As chronic liver diseases, including NASH and alcohol-related cirrhosis, escalate, more patients progress to end-stage liver disease requiring transplantation. Donor scarcity and strict eligibility criteria mean many patients face prolonged wait times, heightening the risk of complications and mortality during this period. This gap has fueled the development and adoption of bridge therapies designed to stabilize patients while they await transplantation.
Bridge therapies encompass a range of interventions, including pharmacological agents that slow fibrosis progression, minimally invasive procedures such as transjugular intrahepatic portosystemic shunt (TIPS) for portal hypertension, and supportive measures to manage complications such as ascites, hepatic encephalopathy, and variceal bleeding. These strategies aim to improve patient survival, optimize liver function, and reduce transplant-related risks. The rising need for bridging solutions is driving innovation in both drug development and procedural techniques, emphasizing individualized treatment plans based on disease severity and comorbidities. Improvements in early diagnosis and monitoring facilitate timely intervention, ensuring that patients remain eligible and stable for transplantation.
Barrier Analysis – High Cost of Advanced Therapies and Limited Access
Innovative treatments, including novel antifibrotic drugs, biologics, and procedural interventions such as liver transplantation, often come with substantial price tags, making them unaffordable for large patient populations, particularly in low- and middle-income regions. Even in developed markets, out-of-pocket expenses, insurance limitations, and complex reimbursement processes can restrict access.
Limited availability of specialized healthcare centers and trained clinicians further constrains treatment delivery, particularly for advanced interventions such as transplant surgery or minimally invasive procedures. Geographic disparities mean that patients in rural or underserved areas may face delays in diagnosis and treatment initiation, worsening disease outcomes. These factors collectively hinder the widespread adoption of advanced therapies, driving a need for cost-effective solutions, equitable healthcare infrastructure, and supportive policies that can improve accessibility and ensure timely management of liver cirrhosis.
Disease Heterogeneity and Late Presentation
Cirrhosis can result from diverse etiologies, including NASH, viral hepatitis, and alcohol-related liver injury, each with distinct progression patterns, complications, and response to therapy. This heterogeneity complicates treatment, as patients may require tailored management strategies addressing both the underlying cause and disease stage. Late presentation is another critical issue, as many patients remain asymptomatic until advanced fibrosis or decompensation occurs.
The time of diagnosis, complications such as portal hypertension, ascites, or hepatic encephalopathy may already be present, limiting therapeutic options and reducing survival rates. Early detection remains difficult due to the silent nature of disease progression, inconsistent screening, and limited patient awareness. These factors collectively drive demand for improved diagnostic tools, personalized treatment approaches, and interventions that can manage complications effectively, making disease heterogeneity and late presentation key considerations shaping the liver cirrhosis treatment market.
Opportunity Analysis – Advancements in Antifibrotic Agents and Combination Regimens
Antifibrotic agents are emerging as a critical component of this strategy, targeting pathways involved in hepatic inflammation, stellate cell activation, and extracellular matrix deposition. By addressing the underlying fibrotic processes, these therapies aim to halt disease progression, reduce complications, and improve long-term outcomes, particularly for patients with NASH or other chronic liver conditions. Combination regimens are gaining prominence as a way to enhance therapeutic efficacy.
Pairing antifibrotic agents with treatments that address metabolic dysfunction, inflammation, or portal hypertension, clinicians can target multiple disease mechanisms simultaneously. This multi-pronged approach is especially valuable for patients with advanced or heterogeneous disease, where single-agent therapies may be insufficient. Ongoing clinical research is refining dosing strategies, safety profiles, and patient selection criteria, allowing for more personalized treatment plans. The integration of antifibrotic therapies into standard care could reduce the need for liver transplantation, improve survival, and enhance quality of life.
Home-Based Complication Management
Managing complications of liver cirrhosis in a home-based setting is becoming an essential strategy to reduce hospitalizations and improve patient quality of life. Cirrhosis often leads to complications such as ascites, hepatic encephalopathy, variceal bleeding, and infections, which traditionally require frequent clinical visits or inpatient care. Home-based management leverages patient monitoring, telemedicine, and caregiver support to detect early warning signs and intervene promptly.
Interventions include home administration of diuretics for ascites control, lactulose or rifaximin for hepatic encephalopathy management, and nutritional counseling to maintain liver function and prevent malnutrition. Remote monitoring tools allow clinicians to track vital signs, fluid status, and lab results, enabling timely dosage adjustments or emergency referrals. Patient education programs help individuals recognize symptoms of decompensation, adhere to medication regimens, and maintain lifestyle modifications, reducing the risk of acute episodes.
Category-wise Analysis
Treatment Type Insights
The antibiotics segment is anticipated to dominate the market, accounting for approximately 52% of the market share in 2026. Its dominance is driven by its critical role in managing cirrhosis-related infections. Patients with advanced liver disease are highly susceptible to bacterial infections, including spontaneous bacterial peritonitis and sepsis, which significantly increase morbidity and mortality. Antibiotics help prevent and treat these infections, reducing complications and hospitalizations. Norfloxacin, a fluoroquinolone antibiotic, is widely employed as a first-line prophylactic treatment to prevent spontaneous bacterial peritonitis (SBP) in patients with cirrhosis and ascites. Regular use of norfloxacin has been shown to significantly reduce the incidence of SBP by limiting bacterial translocation from the gut into the ascitic fluid.
The immunosuppressants segment is likely to be the fastest-growing treatment type, due to the increasing number of liver transplant procedures. Post-transplant patients require immunosuppressive therapy to prevent graft rejection and ensure long-term organ survival. Immunosuppressive agents, including cyclosporine and mycophenolate mofetil, are widely utilized to regulate the immune response while maintaining overall patient safety and health. Advances in drug formulations and monitoring protocols have improved safety and efficacy, encouraging wider adoption. Sanofi manufactures Solupred, a corticosteroid used to reduce inflammation in patients with severe liver inflammation, such as Alcoholic Hepatitis or autoimmune-related liver disorders that may progress to Liver Cirrhosis. Prednisolone helps suppress inflammatory immune responses in the liver, improving liver function and clinical symptoms.
End-user Insights
Hospitals & clinics are expected to dominate the market, contributing nearly 65% of revenue in 2026, as most patients require continuous medical supervision and specialized care. Hospitals and clinics provide comprehensive diagnostic services, including blood tests, imaging, and liver function monitoring, which are essential for managing cirrhosis. They are also the primary centers for administering medications, managing complications such as ascites or infections, and performing advanced procedures, including liver transplantation. The Institute of Liver and Biliary Sciences is a leading healthcare and research center dedicated to the diagnosis, treatment, and research of liver diseases such as Liver Cirrhosis, hepatitis, and liver cancer. It is also known for advanced procedures, including liver transplantation, hepatology care, and liver disease research programs.
Research and academic institutes represent the fastest-growing end-user, supported by their focus on developing novel therapies and understanding disease mechanisms. These institutions conduct preclinical and clinical studies on antifibrotic agents, immunomodulators, and combination regimens, driving innovation in treatment strategies. Their work in identifying biomarkers, optimizing drug dosing, and evaluating efficacy across diverse patient populations accelerates the translation of research into clinical practice. Fibrosis Research Center at the Icahn School of Medicine at Mount Sinai in the U.S., led by Dr. Scott Friedman. This program focuses on understanding liver fibrosis mechanisms and translating discoveries into new diagnostics and therapeutics, including novel antifibrotic compounds that could benefit cirrhosis patients.
Regional Insights
North America Liver Cirrhosis Treatment Market Trends
North America is projected to dominate, accounting for nearly 43% of the global liver cirrhosis treatment market in 2026, driven by increasing awareness of chronic liver diseases and the growing need for early diagnosis and long-term disease management. A major trend in the region is the rising focus on preventive care and routine screening, particularly among individuals at risk due to alcohol consumption, obesity, viral hepatitis, or metabolic disorders. Early detection helps healthcare providers initiate treatment sooner and manage complications more effectively.
Growing use of combination therapies to manage cirrhosis and its complications. Physicians commonly prescribe medications to control portal hypertension, infections, and inflammation while also addressing underlying causes such as viral hepatitis or fatty liver disease. Improved clinical monitoring and treatment protocols are helping patients manage the disease more effectively over time. Technological advancement in healthcare infrastructure is also shaping the market. Hospitals and specialized liver centers in North America increasingly use advanced imaging, laboratory diagnostics, and minimally invasive procedures to evaluate liver damage and guide treatment decisions. In addition, the region is witnessing rising interest in innovative therapies and targeted drugs aimed at slowing disease progression and improving liver function is expanding the target applications for liver cirrhosis treatment.
Europe Liver Cirrhosis Treatment Market Trends
European growth is stimulated by increasing awareness of cirrhosis management benefits, strong regulatory systems, and government-led alcohol-harm and viral-hepatitis programs. Countries such as Germany, France, the U.K., and Italy have well-established hepatology frameworks that support routine liver cirrhosis treatment use and encourage adoption of innovative antibiotic and immunosuppressant delivery methods. These high-efficacy formulations are particularly appealing for hospitals & clinics populations, regulation-conscious centers, and research users, improving survival and coverage rates.
Technological advancements in liver cirrhosis treatment development, such as enhanced rifaximin formulations, application-targeted delivery, and improved antifibrotic grades, are further boosting market potential. European authorities are increasingly supporting research and trials for therapies against both routine and specialized needs, strengthening market confidence. The growing emphasis on convenient, preventive options is aligned with the region’s focus on reducing alcohol-related burden and transplant waitlists. Public awareness campaigns and promotion drives are expanding reach in both hospitals & clinics and research institutes, while suppliers are investing in combination regimens and novel variants to increase efficacy.
Asia Pacific Liver Cirrhosis Treatment Market Trends
Asia Pacific is likely to be the fastest-growing region, driven by rising NASH and viral-hepatitis awareness, increasing government initiatives, and expanding application programs across the region. Countries such as China, India, Japan, and South Korea are actively promoting treatment campaigns to address cirrhosis growth and emerging transplant needs. Liver cirrhosis treatments are particularly attractive in these regions due to their scalable administration, ease of adoption, and suitability for large-scale hospitals & clinics and antibiotic drives in both urban and semi-urban populations.
Technological advancements are supporting the development of stable, effective, and easy-to-administer liver cirrhosis treatments, which can withstand challenging patient profiles and minimize decompensation dependence. These innovations are critical for reaching domestic centers and improving overall liver coverage. Public-private partnerships, increased hepatology expenditure, and rising investment in cirrhosis research and treatment capacity are further accelerating growth. The convenience of treatment delivery, combined with improved control and reduced risk of complications, positions it as a preferred choice.
Competitive Landscape
The global liver cirrhosis treatment market is characterized by dynamic competition between established hepatology leaders and emerging antifibrotic specialists. In North America and Europe, companies such as Gilead Sciences and AbbVie maintain leadership through extensive R&D pipelines, strong ties with liver transplant centers, and robust patient networks. Their innovative antibiotic and immunosuppressant programs address both infection control and post-transplant complications, reinforcing their dominance in advanced therapy segments.
In the Asia Pacific region, local and regional players focus on cost-effective solutions, improving accessibility for a growing patient base, and expanding market penetration in hospitals and clinics. Antibiotic therapies play a pivotal role by preventing infections, reducing hospital readmissions, and enabling integration into standardized care protocols. Strategic collaborations, partnerships, and acquisitions further accelerate portfolio expansion and commercialization, combining clinical expertise with operational scale.
Key Industry Developments:
- In November 2025, Gilead Sciences, Inc. announced new long-term data that reinforced the safety and efficacy profile of Livdelzi® (seladelpar) for people living with primary biliary cholangitis (PBC). The findings showed that Livdelzi maintained consistent efficacy and safety outcomes in patients who switched from obeticholic acid and positively impacted liver stiffness over three years.
- In June 2025, AbbVie announced that the U.S. Food and Drug Administration (FDA) approved a label expansion for MAVYRET® (glecaprevir/pibrentasvir), an oral pangenotypic direct-acting antiviral (DAA) therapy. The FDA approved MAVYRET for the treatment of adults and pediatric patients aged three years and older with acute or chronic hepatitis C virus (HCV) infection, without cirrhosis or with compensated cirrhosis. With this approval, MAVYRET became the first and only DAA therapy authorized to treat patients with acute HCV in eight weeks, achieving a 96% cure rate.
Companies Covered in Liver Cirrhosis Treatment Market
- Gilead Sciences
- AbbVie
- Bristol-Myers Squibb
- Merck & Co.
- Novartis
- Roche
- Amgen
- Eisai
- Hepion Pharmaceuticals
Frequently Asked Questions
The global liver cirrhosis treatment market is projected to reach US$16.1 billion in 2026.
Increasing cases of NASH, viral hepatitis, and alcohol-related cirrhosis are driving demand for effective treatment options, including antibiotics, immunosuppressants, and antifibrotic therapies.
The liver cirrhosis treatment market is poised to witness a CAGR of 7.1% from 2026 to 2033.
Development of antifibrotic agents, novel immunosuppressants, and personalized combination treatments presents opportunities for improved patient outcomes and expansion in both developed and emerging markets.
Gilead Sciences, AbbVie, Novartis, Merck & Co., and Bristol-Myers Squibb are the key players.













