- Pharmaceuticals
- Fatty Acid Oxidation Disorder (FAOD) Market
Fatty Acid Oxidation Disorder (FAOD) Market Size, Share, and Growth Forecast, 2026 - 2033
Fatty Acid Oxidation Disorder (FAOD) Market by Treatment Type (Levocarnitine, Triheptanoin, Dextrose, Riboflavin), Route of Administration (Oral, Intravenous, Intranasal), Distribution Channel (Hospital Pharmacies, Online Pharmacies, Retail Pharmacies), and Regional Analysis for 2026-2033
Fatty Acid Oxidation Disorder (FAOD) Market Share and Trends Analysis
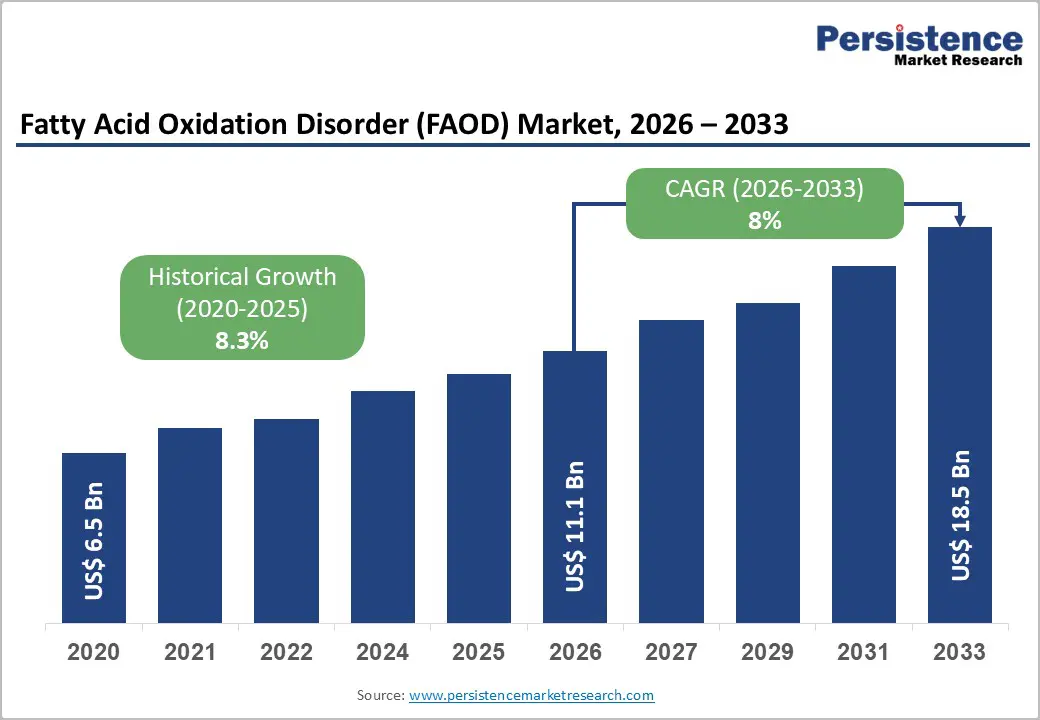
The global fatty acid oxidation disorder market size is likely to be valued at US$ 11.1 billion in 2026, and is projected to reach US$ 18.5 billion by 2033, growing at a CAGR of 8% during the forecast period 2026−2033.
The prime growth driver for this market is the increasing frequency of identification of FAODs through improved newborn screening programs and broader metabolic testing protocols. According to the National Institutes of Health (NIH), these inherited metabolic conditions affect a notable proportion of live births worldwide, underscoring the need for structured therapeutic pathways. Hospitals and specialty clinics are therefore integrating early detection tools with dietary management plans and pharmacological interventions to reduce complications and improve patient outcomes.
Technological and therapeutic innovation is further shaping this market landscape. Expanded genetic sequencing capabilities are enabling precise identification of enzyme deficiencies associated with FAODs, which is supporting tailored treatment strategies. Regulatory approvals for novel therapies are enhancing available care options, while biopharmaceutical companies are increasing research and development investments in rare metabolic disease portfolios. Clinicians are also gaining greater awareness of symptom variability, which is improving referral accuracy and continuity of care.
Key Industry Highlights
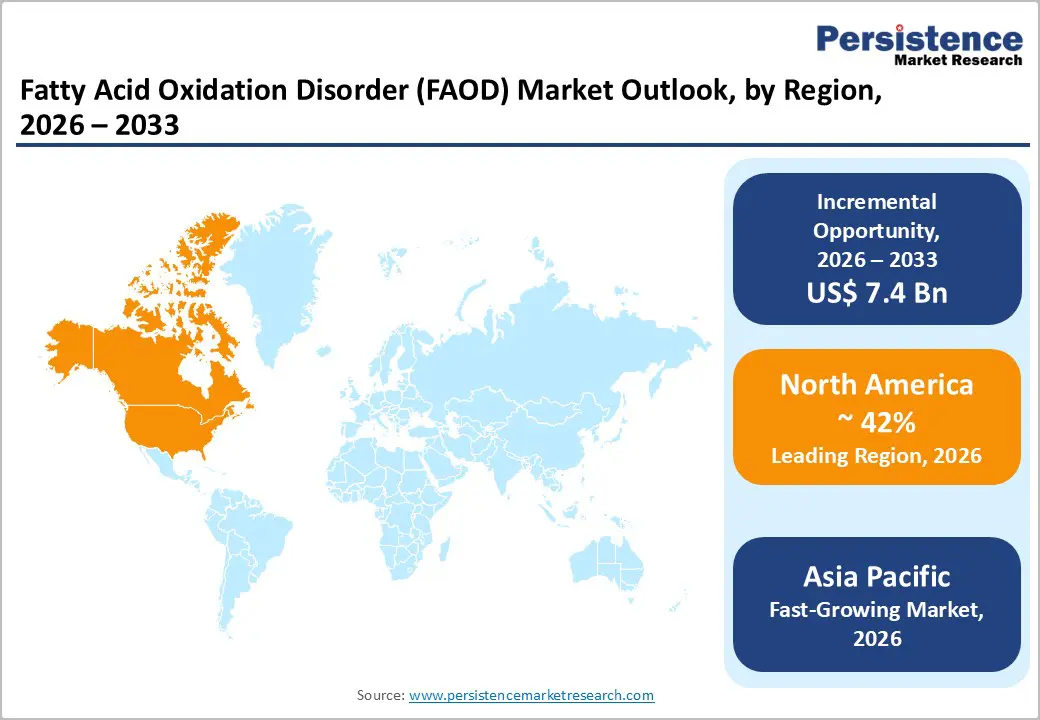
- Dominant Region: North America is expected to command about 42% market share in 2026, supported by advanced healthcare systems featuring specialized centers for metabolic conditions.
- Fastest-growing Market: The Asia Pacific market is slated to be the fastest-growing through 2033, driven by increasing healthcare investments and rising awareness of genetic disorders.
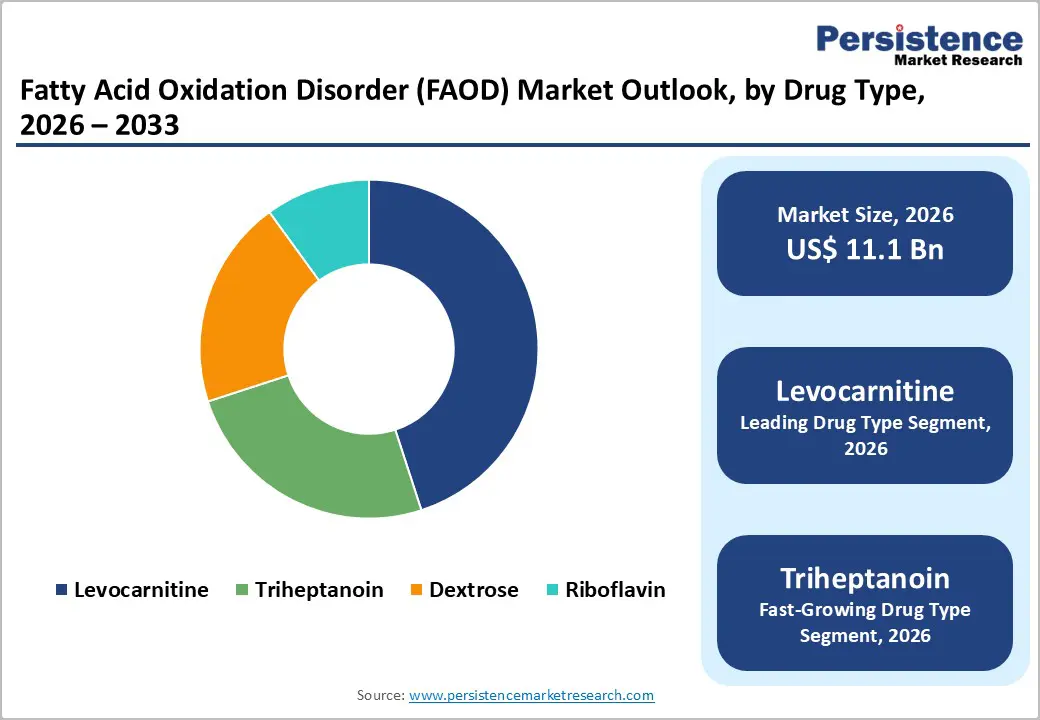
- Leading & Fastest-growing Treatment Type: Levocarnitine is poised to command approximately 45% revenue share in 2026, while triheptanoin is likely to be the fastest-growing segment during the 2026-2033 forecast period.
- Leading & Fastest-growing Distribution Channel: Hospital pharmacies are anticipated to lead with about 45% revenue share in 2026, with online pharmacies registering the highest CAGR between 2026 and 2033.
- Key Driver: Newborn screening programs and orphan drug regulations are driving higher diagnosis rates and innovative FAOD treatments worldwide.
- Major Opportunity: Gene therapy advancements and precision biomarkers are generating opportunities for curative, patient-tailored solutions in the FAOD space.
| Key Insights | Details |
|---|---|
| Fatty Acid Oxidation Disorder (FAOD) Market Size (2026E) | US$ 11.1 Bn |
| Market Value Forecast (2033F) | US$ 18.5 Bn |
| Projected Growth (CAGR 2026 to 2033) | 8% |
| Historical Market Growth (CAGR 2020 to 2025) | 8.3% |
Market Factors – Growth, Barriers, and Opportunity Analysis
Rising Prevalence and Increased Disease Awareness
Healthcare professionals and patient advocacy groups are strengthening awareness of fatty acid oxidation disorders through coordinated outreach and education initiatives. Organizations such as the National Organization for Rare Disorders (NORD) and the FOD Family Support Group are conducting educational programs, facilitating fundraising campaigns, and building peer support communities for affected families. These efforts are equipping caregivers with practical resources and are fostering informed disease management. Major academic medical centers are establishing specialized metabolic clinics that are integrating genetics, clinical nutrition, neurology, and metabolic medicine. This multidisciplinary model is enabling coordinated care pathways and personalized treatment planning based on individual biochemical profiles.
Pharmaceutical companies and professional societies are training clinicians to recognize FAOD symptoms promptly, which is reducing diagnostic delays and improving referral accuracy. As clinical recognition is increasing, patient identification rates are rising, thereby expanding demand for targeted therapeutics and structured dietary interventions. For industry stakeholders, this trend is indicating a stable and growing patient base that supports long term investment in rare metabolic care. Companies that are forming strategic collaborations with advocacy networks are gaining actionable insights into real world patient needs, which is informing product design and access strategies. Sustained awareness building is therefore reinforcing a supportive ecosystem that is improving health outcomes while creating pathways for innovation and responsible market expansion.
Clinical Heterogeneity and Limited Treatment Options
FAODs are presenting significant clinical heterogeneity, which is complicating standardized treatment pathways. Symptom severity and metabolic stability are varying widely across patients, even within the same genetic subtype. Clinicians are therefore tailoring interventions to individual biochemical and clinical profiles, which is increasing care complexity and resource utilization. Current management strategies are focusing on structured dietary control, strict avoidance of prolonged fasting, and targeted supplementation such as levocarnitine, dextrose, and riboflavin. Triheptanoin is being used specifically for long chain fatty acid oxidation disorders (LC-FAODs), while many other subtypes continue to rely on supportive therapy and rapid response during metabolic decompensation episodes.
Validated biomarkers for monitoring therapeutic response remain limited, which is constraining optimization of long term management strategies. Several emerging treatments are still lacking extensive longitudinal data, and recurrent metabolic crises are leading to hospital admissions that burden families and healthcare systems. For industry leaders, these constraints are signaling clear unmet clinical needs. Companies that are investing in biomarker discovery, precision medicine platforms, and gene based therapeutic approaches are positioning themselves to address underlying metabolic defects rather than symptoms alone. Early collaboration with metabolic specialists and research centers is strengthening clinical development pathways and facilitating regulatory engagement. Strategic emphasis on personalization and durable efficacy is expected to support improved patient outcomes while enabling sustainable value creation in this expanding rare disease segment.
Gene Therapy and Precision Medicine Development
Gene therapy innovation is reshaping the treatment paradigm for FAODs by targeting underlying enzymatic defects rather than managing symptoms alone. Biotechnology companies are developing candidates that deliver functional genes through viral vectors to correct specific metabolic deficiencies within defined FAOD subtypes. These programs are aiming to provide durable therapeutic benefit instead of lifelong supportive care. Developers are also leveraging gene editing technologies such as clustered regularly interspaced short palindromic repeats (CRISPR) to enable precise genomic modification. Precision medicine approaches are aligning treatment selection with individual mutation profiles through pharmacogenomic analysis, which is allowing clinicians to design intervention strategies based on detailed genetic characterization.
From a strategic standpoint, companion diagnostics and validated biomarker platforms are becoming essential components of this evolving ecosystem. Genetic testing is integrating with digital health monitoring tools to track metabolic stability and adjust therapeutic regimens in real time. Companies that are collaborating with academic research centers and metabolic specialists are accelerating clinical development while aligning with regulatory expectations for safety and long term efficacy. Early investment in scalable gene therapy platforms is enabling broader applicability across multiple FAOD subtypes and strengthening competitive positioning. Organizations that are prioritizing innovation, regulatory readiness, and cross sector partnerships are likely to establish sustained leadership as demand for transformative rare disease therapies continues to rise.
Category-wise Analysis
Drug Type Insights
Levocarnitine is anticipated to hold approximately 45% of the fatty acid oxidation disorder market revenue share in 2026, as it serve as a foundational therapy across FAOD subtypes. This amino acid derivative is facilitating the transport and excretion of excess fatty acids while supporting restoration of coenzyme A balance in patients with metabolic impairment. Clinicians are prescribing it routinely in primary carnitine deficiency and are using it as adjunctive therapy in broader FAOD management protocols. Its established safety record, availability in both oral and intravenous formulations, and widespread generic accessibility are reinforcing its clinical acceptance. A strong evidence base and long term familiarity among metabolic specialists are further sustaining its market position.
Triheptanoin is expected to be the fastest-growing segment during the 2026-2033 forecast period, given that it is the first therapy specifically approved by the U.S. Food and Drug Administration (FDA) for long LC-FAODs. This synthetic medium chain triglyceride composed of three seven carbon fatty acids is providing an alternative energy substrate that bypasses defective long chain oxidation pathways. Its anaplerotic mechanism is addressing metabolic insufficiency at a biochemical level rather than offering only symptomatic support. Physician adoption is increasing as clinical experience grows, and reimbursement coverage is expanding across additional regions. Ongoing research into broader FAOD subtypes is likely to strengthen its therapeutic footprint and reinforce its differentiated value proposition within this specialized market.
Route of Administration Insights
The oral route is likely to remain dominant in 2026, since it supports long-term disease control with high patient adherence and practical convenience. Oral formulations are capturing the majority of revenue share as patients and caregivers are favoring home based therapy over facility dependent options. Available formats such as tablets, capsules, liquid suspensions, and chewable preparations are accommodating both pediatric and adult populations with varying clinical needs. For individuals living with fatty acid oxidation disorders, lifelong management is requiring consistent supplementation and metabolic stabilization. Oral therapy is enabling structured chronic care without dependence on specialized infusion centers or trained personnel, thereby reducing overall healthcare utilization and improving continuity of treatment.
Intravenous administration is expected to record the fastest growth between 2026 and 2033, as it plays a critical role in emergency metabolic management. During episodes of metabolic decompensation triggered by infection, prolonged fasting, or physiological stress, rapid intravenous infusion of concentrated dextrose and levocarnitine is correcting hypoglycemia and suppressing abnormal fatty acid metabolism. Hospitals are strengthening preparedness protocols for acute FAOD crises, and specialized metabolic clinics are enhancing infusion capabilities to deliver timely intervention. Expanded hospital pharmacy infrastructure and improved reimbursement pathways for emergency care are supporting adoption of intravenous formulations. Healthcare systems that are integrating clear escalation protocols between oral maintenance therapy and acute intravenous rescue strategies will have strengthened patient safety frameworks and optimized clinical outcomes across this complex rare disease population.
Distribution Channel Insights
Hospital pharmacies are slated to dominate with around 45% of the FAOD market revenue share in 2026. Specialized medical centers are anchoring diagnosis, therapy initiation, and emergency stabilization for fatty acid oxidation disorders. Several metabolic disorder programs are operating within academic hospitals and pediatric institutions, where multidisciplinary teams are coordinating genetics, nutrition, neurology, and metabolic medicine services. Hospital pharmacies are managing specialty drugs that require controlled storage, prior authorization processing, and alignment with standardized clinical pathways. Prescription volumes are increasing during inpatient admissions, emergency department interventions, and structured outpatient follow up visits. Integrated hospital based oversight is ensuring dose adjustment, safety monitoring, and structured caregiver education at the point of therapy initiation.
Online pharmacies are projected to be the fastest-growing distribution channel through 2033, spurred by the speedy digitalization of healthcare delivery, with a deepening focus on making it patient-centric. Electronic pharmacy platforms are enabling round the clock ordering, competitive pricing structures, automated refill reminders, and direct home delivery. These advantages are aligning well with chronic disease populations that require consistent long term medication access, including individuals living with FAODs. Regulatory authorities are strengthening verification and authentication systems to ensure product integrity and patient safety within digital channels. As reimbursement pathways and telehealth integration are advancing, online distribution models are complementing hospital based care and enhancing accessibility. Organizations that are integrating digital dispensing solutions with clinical oversight frameworks will have strengthened continuity of care while improving operational efficiency in this evolving distribution landscape.
Regional Insights
North America Fatty Acid Oxidation Disorder (FAOD) Market Trends
North America is projected to account for approximately 42% of the fatty acid oxidation disorder market share in 2026, boosted primarily by the United States. Comprehensive newborn screening programs operating across all states are enabling early identification of metabolic abnormalities, which is strengthening timely intervention. Advanced healthcare infrastructure includes specialized metabolic centers that coordinate multidisciplinary care. Favorable reimbursement systems are covering high value rare disease therapies, which is improving patient access. The FDA is reinforcing innovation through orphan drug incentives and expedited review pathways. Recent commercial launches, including triheptanoin following regulatory approval, are further stimulating therapeutic adoption, while enhanced screening protocols are increasing diagnostic rates.
Ongoing medical education initiatives are raising clinician awareness and refining referral accuracy. United States biotechnology companies are investing heavily in gene therapy pipelines and precision medicine platforms targeting inherited metabolic conditions. Established players such as Ultragenyx Pharmaceutical and Pfizer are competing alongside emerging biotechnology firms, creating a dynamic innovation environment supported by active venture capital funding. Companies that are partnering with advocacy organizations and leveraging robust clinical trial networks are accelerating evidence generation and regulatory alignment. Strategic focus on localized commercialization models within high expenditure healthcare systems will have strengthened long term positioning as the diagnosed patient population continues to expand.
Europe Fatty Acid Oxidation Disorder (FAOD) Market Trends
Europe is foreseen to emerge as the second-largest market for fatty acid oxidation disorders in 2026, powered by structured public healthcare systems and coordinated rare disease frameworks. Germany, the U.K., France, Spain, and Italy are anchoring regional demand through established metabolic screening programs and academic research capacity. Germany is operating mature newborn screening infrastructure and research driven clinical networks supported by comprehensive insurance coverage. The United Kingdom is leveraging specialized rare disease services within the National Health Service (NHS), while France is implementing national rare disease strategies linked to designated reference centers. At the regulatory level, the European Medicines Agency (EMA) is granting orphan designations that are encouraging therapeutic innovation and facilitating coordinated market access across European Union (EU) member states.
Collaborative clinical ecosystems are further strengthening the region’s position. The European Reference Network for Hereditary Metabolic Disorders is pooling expertise to enhance diagnostic accuracy and standardize care pathways across borders. Companies are benefiting from centralized regulatory procedures that enable simultaneous access to multiple national markets following EMA approval. Public private partnerships supported by European Union research funding programs are accelerating translational science and early phase development. Regional firms such as Recordati Rare Diseases are competing alongside multinational entrants, creating a balanced competitive landscape. Organizations that are aligning with academic networks, securing participation in collaborative trials, and engaging early with regulatory authorities will have strengthened their strategic foothold as patient identification and therapeutic demand continue to rise.
Asia Pacific Fatty Acid Oxidation Disorder (FAOD) Market Trends
Asia Pacific is poised to become the fastest-growing regional market for FAODs from 2026 to 2033, driven by improving healthcare infrastructure and supportive policy initiatives. China, Japan, India, South Korea, and ASEAN membersare strengthening diagnostic capabilities and expanding access to specialized metabolic care. Growing middle class populations are gaining broader insurance coverage, and governments are prioritizing rare disease frameworks within national health agendas. Urban centers are scaling newborn screening programs and enhancing clinician training in inherited metabolic disorders. At the same time, pharmaceutical manufacturing capacity is increasing across the region, supported by established active pharmaceutical ingredient (API) production ecosystems and cost competitive supply chains.
Regional industrial expansion is also reinforcing market attractiveness. Contract development and manufacturing organizations (CDMOs) are supporting global pharmaceutical pipelines through integrated development and production services. China is implementing national rare disease reimbursement pathways, while Japan is advancing ultra-orphan incentives to accelerate approvals. India is building biotechnology capabilities that enable affordable therapeutic alternatives. International firms are forming partnerships with local stakeholders to navigate regulatory pathways and access specialized talent pools. Strategic prioritization of clinical trial sites in high growth hubs is enabling diverse patient recruitment and faster data generation. Companies that are investing in physician education, early diagnosis campaigns, and locally adapted commercialization models will have strengthened long term positioning as healthcare modernization continues across the region.
Competitive Landscape
The global fatty acid oxidation disorder market structure is moderately consolidated, with a small group of specialized and multinational companies shaping competitive direction. Prominent participants include Ultragenyx Pharmaceutical, Sigma-Tau Pharmaceuticals, and Pfizer. These organizations are advancing research, regulatory submissions, and global commercialization strategies for targeted FAOD therapies. Market leaders are expanding portfolios through development of novel treatments, lifecycle management initiatives, and selective geographic expansion. Partnerships with academic centers and rare disease networks are strengthening clinical validation and supporting integrated patient care frameworks as diagnosis rates continue to rise.
Competitive intensity is driving innovation across therapeutics and diagnostics. Companies are investing in gene based platforms, metabolic modulators, and precision testing tools that address heterogeneity and long term disease control challenges. Strategic differentiation is increasingly centered on pipeline depth, orphan drug positioning, and collaboration models that accelerate regulatory approval. Executives who are systematically monitoring competitor pipelines, licensing agreements, and clinical trial progress will be better positioned to anticipate market shifts. Agile firms that align product development with evolving screening programs and patient advocacy efforts are likely to capture incremental share and sustain long term value creation within this expanding rare disease segment.
Key Industry Developments
- In February 2026, Candriam S.C.A. significantly increased its investment in Ultragenyx Pharmaceutical Inc. by purchasing additional shares and raising its overall stake, signaling confidence from an institutional investor in the company’s long-term prospects. This move comes amid broader analyst coverage that generally supports the stock’s outlook, even as legal risks and market volatility remain under consideration.
- In December 2025, researchers from Novo Nordisk Research Centre Oxford developed a high-throughput fluorescence-based assay using quinone methide probes to quantify fatty acid oxidation (FAO) in human skeletal muscle cells, addressing limitations in traditional methods for studying obesity and type 2 diabetes.
Companies Covered in Fatty Acid Oxidation Disorder (FAOD) Market
- Ultragenyx Pharmaceutical Inc.
- Sigma-Tau Pharmaceuticals
- Northeast Pharmaceutical Group Co., Ltd.
- Pfizer Inc.
- Leadiant Biosciences, Inc.
- Merck KGaA
- Bedford Laboratories
- Reneo Pharmaceuticals, Inc.
- Mylan Laboratories Limited
- Daiichi Sankyo Ltd.
- Biosynth AG
- AminoScience LLC
- Chengda Pharmaceuticals Co., Ltd.
- Spectrum Chemical Manufacturing Corporation
- Lonza Group Ltd.
Frequently Asked Questions
The global fatty acid oxidation disorder (FAOD) market is projected to reach US$ 11.1 billion in 2026.
Newborn screening expansions, novel therapy approvals, and R&D funding and therapy approvals, such as triheptanoin, are driving market growth.
The market is poised to witness a CAGR of 8% from 2026 to 2033.
Major opportunities lie in gene therapies, precision medicine, personalized medicine, and biomarkers that enable targeted treatments.
Ultragenyx Pharmaceutical Inc., Sigma-Tau Pharmaceuticals, and Pfizer Inc. are some of the key players in the market.








