
- Pharmaceuticals
- Enzyme Replacement Therapy Market
Enzyme Replacement Therapy Market Size, Share, Growth, and Regional Forecast, 2026 to 2033
Enzyme Replacement Therapy Market by Therapeutic Conditions (Mucopolysaccharidosis (MPS), Gaucher Disease, Fabry Disease, Pompe Disease, Lysosomal Acid Lipase Deficiency, Others), by Route of Administration, by Distribution Channel, and by Regional Analysis, from 2026 to 2033
Enzyme Replacement Therapy Market Share and Trends Analysis
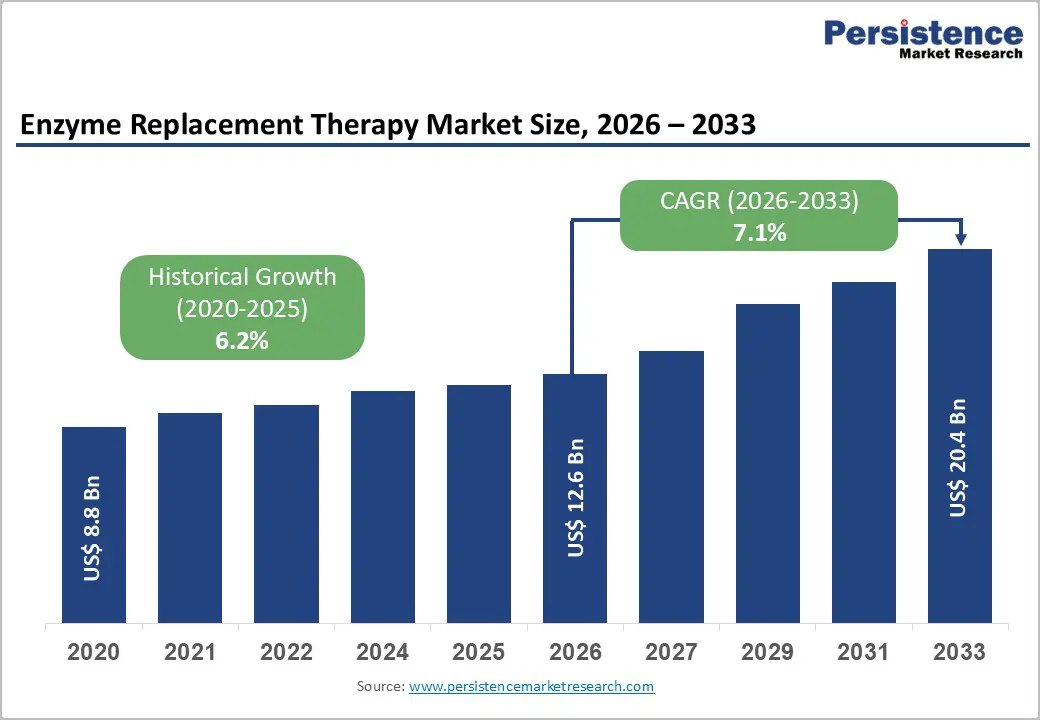
The global enzyme replacement therapy market size is expected to be valued at US$ 12.6 billion in 2026 and projected to reach US$ 20.4 billion by 2033, growing at a CAGR of 7.1% between 2026 and 2033.
Innovations in biotechnology and enzyme manufacturing techniques have facilitated the development and approval of new therapies, thereby expanding access to care. Regulatory support in developed nations and rising diagnostic rates also contributed to market growth. However, challenges such as the high costs of therapy, limited availability in certain regions, and strict regulatory hurdles posed constraints that hindered widespread access and adoption.
Key Industry Highlights
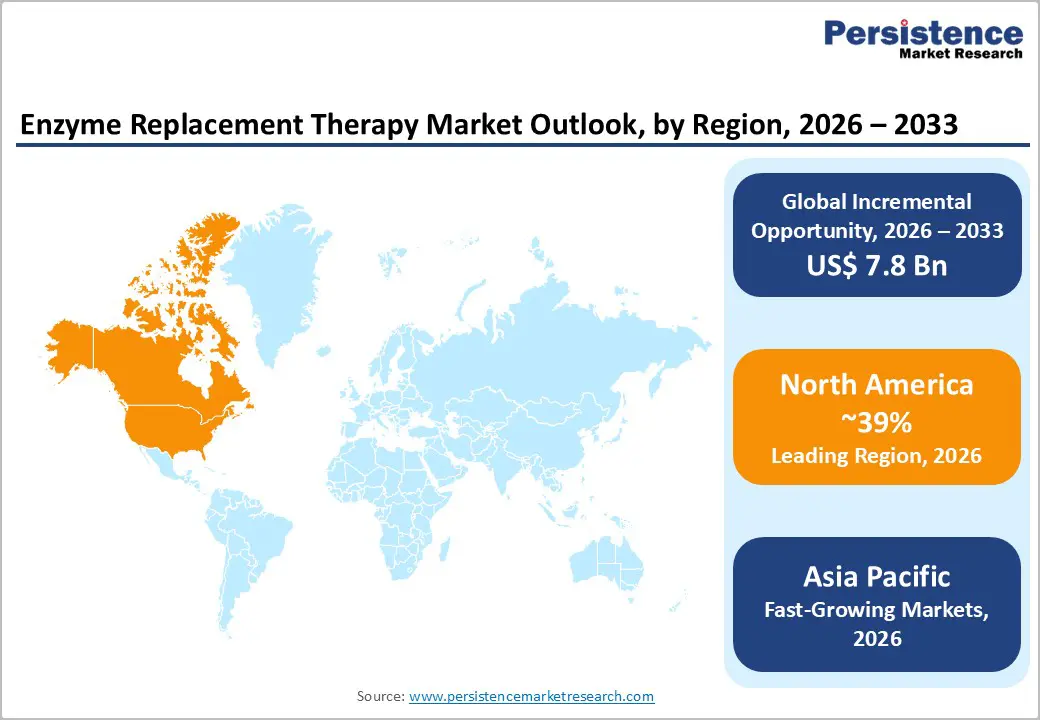
- Leading Region: North America leads due to high rare disease diagnosis rates, advanced biologics infrastructure, strong reimbursement, and the presence of major ERT manufacturers.
- Fastest Growing Region: Asia Pacific is the fastest growing, driven by improving diagnostic capabilities, rising awareness of lysosomal storage disorders, healthcare spending growth, and government support.
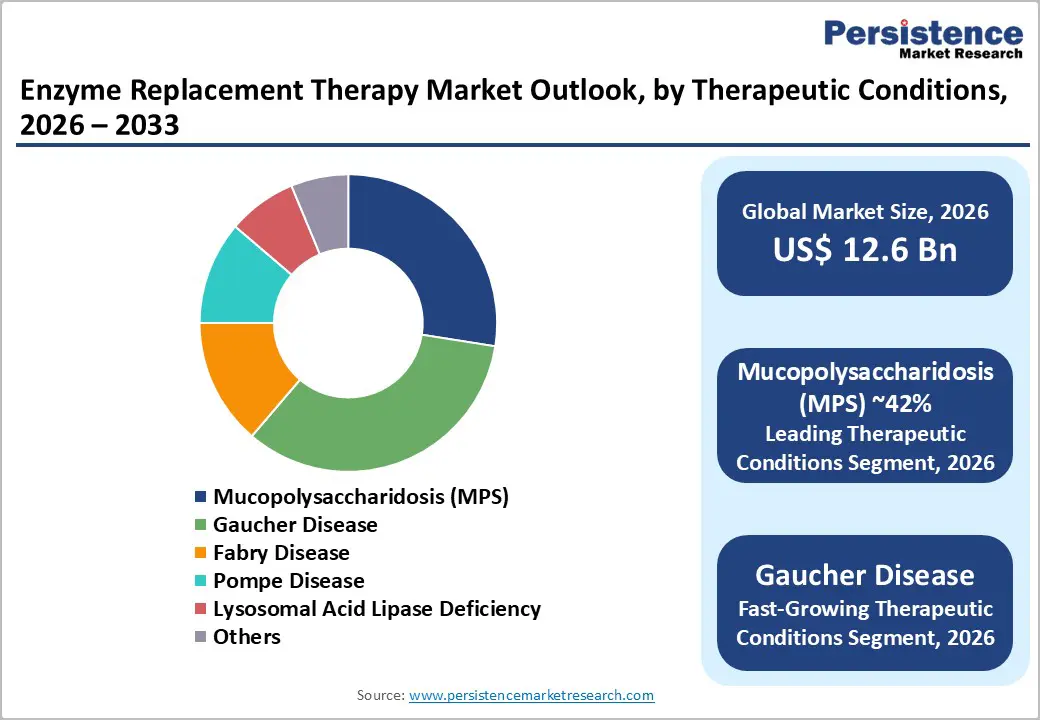
- Dominant Segment: Mucopolysaccharidosis (MPS) is dominant, supported by early pediatric diagnosis, lifelong therapy requirements, higher treatment intensity, and sustained patient adherence.
- Fastest-Growing Segment: Gaucher disease is the fastest-growing segment, driven by improved screening, expanded access to therapy, rising awareness, and favorable long-term treatment outcomes.
| Global Market Attributes | Key Insights |
|---|---|
| Enzyme Replacement Therapy Market Size (2026E) | US$ 12.6 Bn |
| Market Value Forecast (2033F) | US$ 20.4 Bn |
| Projected Growth (CAGR 2026 to 2033) | 7.1% |
| Historical Market Growth (CAGR 2020 to 2024) | 6.2% |
Market Dynamics
Driver - Rising Prevalence of Rare Diseases to Propel Market Growth
The global prevalence of various lysosomal storage disorders, including Gaucher, Fabry, Pompe, and MPS, is steadily rising. According to data provided by the National Institute of Neurological Disorders and Stroke, around 1 in 40,000 individuals in the U.S. were living with Pompe Disease, which was believed to be around 33000 cases.
According to the online studies, nearly 11.6 million infants were examined for Pompe Disease across eight countries and four continents. A ratio of 1 to 18,711 newborns was identified as suffering from the disease. The presence of a substantial patient population afflicted by rare lysosomal storage disorders creates an increased demand for effective therapeutic alternatives, including enzyme replacement therapy. Enhanced initiatives by governments and non-profit organizations across several nations to raise public awareness contribute to a progressive increase in the detection rate of rare diseases.
Critical elements, along with advantageous reimbursement policies and government funding for treatment in developed nations, are enhancing the acceptance rate of this therapy. It is expected to drive market expansion over the forecast period.
Innovations in Biotechnological and Pharmaceutical Techniques Push Demand
Continuous innovations in biotechnology and pharmaceutical manufacturing processes are crucial to the growth of the ERT industry. Enhanced enzyme production techniques, such as recombinant DNA technology, allow for more efficient and consistent production of therapeutic enzymes. Innovations in delivery systems, including nanoparticles and encapsulation methods, have improved enzyme stability and bioavailability, allowing for more targeted delivery. Such innovations reduce the number of doses required, enhance therapeutic efficacy, and minimize side effects.
By increasing the accessibility and effectiveness of enzyme replacement therapies, these biotechnological improvements make ERT a more viable treatment option, contributing to market expansion. Several companies are seeking expedited approvals from government bodies to distribute their products. For example, in November 2023, Takeda received the U.S. FDA approval for Adzynma, a recombinant ADAMTS13 enzyme replacement therapy for congenital thrombotic thrombocytopenic purpura (cTTP).
Restraints - High Costs of Enzyme Replacement Therapies to Limit Demand
One of the primary restraints on the growth of the enzyme replacement therapy market is the high cost associated with these treatments. ERT typically requires complex biotechnological processes to produce therapeutic enzymes, particularly for the treatment of rare and genetic diseases. The intricate production process results in high manufacturing costs, which are ultimately borne by patients and healthcare systems.
For patients in low to middle-income countries, accessing ERT can be financially prohibitive, even with health insurance. This cost burden on healthcare providers and insurers can limit the accessibility and coverage for such therapies, creating a significant barrier to market expansion. Additionally, this creates hurdles to developing cost-effective biosimilars or alternative funding models, slows market growth, and restricts access to life-saving treatments.
Opportunity - Development of Oral Enzyme Replacement Formulations to Foster Opportunities
Oral formulations of ERT represent a transformative growth opportunity, particularly as research continues to identify strategies to protect therapeutic enzymes from degradation in the gastrointestinal tract. Traditionally, ERTs are administered via injections or infusions, which can be uncomfortable and inconvenient for patients.
Oral formulations, however, could significantly improve patient compliance and quality of life by allowing self-administration at home. Innovations in encapsulation and nanoparticle technology could help enzymes survive the digestive process, making oral ERT a feasible option for a wide range of diseases. Pharmaceutical companies are exploring these innovative delivery methods, which, if successful, would revolutionize the administration of ERT. This is anticipated to make it simpler and more accessible for patients globally. For instance, since early 2023, Protalix BioTherapeutics has been accelerating OPRX-106, an oral enzyme therapy. It comprises a plant-cell-expressed recombinant human tumor necrosis factor receptor II fused to an IgG1 Fc domain.
Category-wise Analysis
By Therapeutic Conditions Insights
Based on therapeutic conditions, the market is divided into enzyme replacement therapy for Fabry disease, Gaucher disease, mucopolysaccharidosis (mps), Pompe disease, and lysosomal acid lipase deficiency. Out of these, the Gaucher disease segment dominates the market. ERT for Gaucher disease held the leading market share in 2025, driven by rising prevalence and awareness of the disease, resulting in more patients being diagnosed and treated. Enzyme Replacement Therapy (ERT), particularly with imiglucerase, has become a primary treatment, significantly alleviating symptoms and improving patients' quality of life.
Innovations in biotechnology have improved the production and distribution of these medicines, increasing their accessibility. Robust backing from healthcare systems and continuous research into novel treatment alternatives significantly enhance market expansion.
By Route of Administration Insights
Based on the route of administration, the market is classified into oral and injectable enzyme replacement therapy. Among these, the injectable enzyme replacement therapy segment dominates the market. It is due to the rapid absorption and effective delivery of therapeutic enzymes directly into the bloodstream, which is important for treating conditions like lysosomal storage disorders.
Injectables enable enzymes to bypass the digestive tract and are administered directly into the bloodstream, resulting in rapid and efficient systemic absorption. It is particularly beneficial for ERT, as several target diseases, such as lysosomal storage disorders, require enzymes to reach cells throughout the body. Injectable administration also provides nearly complete bioavailability, ensuring that the full therapeutic dose reaches the target cells and tissues. This efficiency is important in enzyme therapy, where each enzyme dose is expensive and precise dosing is critical.
New formulations, such as enzyme-loaded nanoparticles and liposomes, have been developed for injectable use to enhance targeting and delivery to specific tissues and organs. Innovations in the formulation of injectable enzymes can reduce the likelihood of side effects and improve patient comfort, addressing a common concern associated with injections.
Regional Insights
North America Enzyme Replacement Therapy Market Trends
North America's enzyme replacement therapy market dominated the global market and accounted for the largest revenue share of 39% in 2025. It is attributed to a high prevalence of rare genetic disorders and a robust healthcare infrastructure. The region benefits from significant investments in research and development, fostering innovation in ERT products. In addition, supportive government policies and funding initiatives enhance access to unique therapies. Leading pharmaceutical companies and specialized treatment centers strengthen the market, ensuring that patients receive timely and effective treatments for conditions such as Gaucher and Fabry.
The enzyme replacement therapy market in the U.S. dominates North America, and it accounted for the most significant revenue share in 2025. It is driven by a strong emphasis on patient-centric approaches and advanced medical technologies. High healthcare expenditure facilitates access to cutting-edge treatments, while extensive insurance coverage supports patient affordability.
Growing awareness of rare diseases among healthcare professionals and the public leads to earlier diagnosis and intervention, thereby increasing demand for ERT. The country's commitment to improving rare disease management through regulatory support and innovative clinical trials contributes significantly to the growth of the regional market.
Asia Pacific Enzyme Replacement Therapy Market Trends
The Asia-Pacific enzyme replacement therapy (ERT) market is experiencing rapid growth, driven by rising diagnoses of rare diseases, increasing healthcare expenditure, and improved access to advanced treatments. The region’s market size is forecast to grow significantly through the end of the decade, with estimates showing double-digit compound annual growth rates as healthcare systems in China, India, and Southeast Asia enhance diagnostic and treatment capabilities. Japan and China remain key contributors due to robust research infrastructure, regulatory support for novel therapies, and expanded insurance coverage for rare genetic disorders. Rising awareness of lysosomal storage diseases such as Gaucher and Pompe, along with technological advancements in drug formulation and delivery, further stimulate adoption of ERT products. Hospitals and specialty clinics continue to dominate treatment delivery, while collaborations between global pharmaceutical companies and local stakeholders are fostering product development and market penetration across the Asia Pacific region.
Competitive Landscape
The enzyme and collagen replacement therapy industry is highly competitive, with a mix of established pharmaceutical giants and emerging biotech firms. Key players like Sanofi, Takeda, and Pfizer dominate due to their strong research and development capabilities, extensive product portfolios, and global distribution networks. They mainly focus on extending indications for ERT products and developing novel formulations, including oral and gene therapy-based alternatives.
Emerging firms like Protalix BioTherapeutics and Anagram Therapeutics are innovating with unique delivery technologies, aiming to improve patient compliance and treatment accessibility. Strategic partnerships, mergers, and acquisitions are common, allowing companies to enhance their presence and leverage unique biotechnological techniques. Intense competition encourages innovation, driving the market’s growth while meeting the rising demand for effective and convenient ERT solutions.
Key Industry Developments:
- In January 2024, the U.S. Food and Drug Administration (FDA) classed JCR Pharmaceuticals' enzyme replacement medication, JR-441, as an orphan medicine for Sanfilippo syndrome type A.
- In August 2023, Amicus Therapeutics Inc. based in Philadelphia, introduced Pombiliti for late-onset Pompe disease in the U.K. The Medicines and Healthcare products Regulatory Agency (MHRA) approved it. The treatment combines miglustat with cipaglucosidase alfa to treat symptoms and raise the patient's quality of life.
Companies Covered in Enzyme Replacement Therapy Market
- Sanofi Genzyme
- Shire
- BioMarin Pharmaceutical Inc.
- Amicus Therapeutics
- Alexion Pharmaceuticals
- Ultragenyx Pharmaceutical Inc.
- Sarepta Therapeutics
- Biomarin
- Genzyme (now part of Sanofi)
- Takeda Pharmaceuticals
- Pfizer Inc.
- AbbVie Inc.
- Vertex Pharmaceuticals
- Sangamo Therapeutics
- Others
Frequently Asked Questions
The global enzyme replacement therapy market is projected to be valued at US$ 12.6 Bn in 2026.
Rising prevalence of rare genetic disorders, improved diagnosis, expanding treatment access, favorable reimbursement, and continuous innovation in biologic drug development.
The global market is expected to witness a CAGR of 7.1% between 2026 and 2033.
The average annual cost of ERT ranges between US$ 40000 to US$ 60000 per patient.
North America is the leading region in the global enzyme replacement therapy market.







