![]() 231 Users Online
231 Users Online
Orally Disintegrating Tablet Market Segmented By Anti-Psychotics, Anti-Epileptics, CNS Stimulants, Anxiolytics, Anti-Parkinsonian Drugs, Anti-Hypertensives, NSAIDS, Anti-Allergy Drugs, Proton Pump Inhibitors Drug Class with CNS Diseases, GI Diseases, CVS Disorders, Allergy Disease Indication
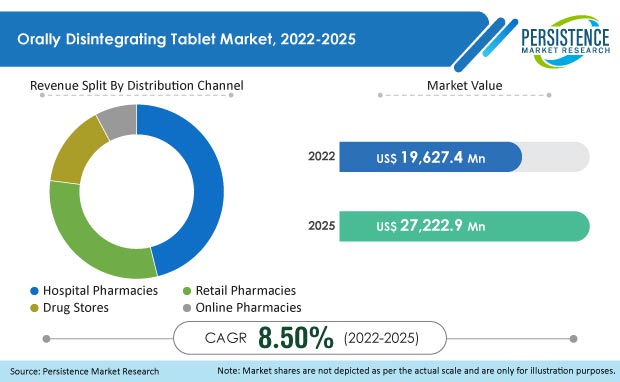
The Global Orally Disintegrating Tablet Market value is projected to exceed US$ 19,627.4 million by the year 2025 at a CAGR of 8.5%. The oral route of drug administration is the usual approach in the pharmaceutical business since it is the most cost-effective, easier, and safer way of drug administration. The tablets are normally taken with water.
The oral tablets enter the gastrointestinal tract and are immediately absorbed into the bloodstream. However, the orally disintegrating tablets are solid dosage forms that quickly dissolve in the mouth and do not require water to ingest. Orally disintegrating tablets are increasingly being used as an alternative to traditional tablets or capsules.
| Attribute | Key Insights |
|---|---|
|
Orally Disintegrating Tablet Market Size (2022) |
US$ 19,627.4 Mn |
|
Projected Market Value (2025) |
US$ 27,222.9 Mn |
|
Global Market Growth Rate (2022-2025) |
8.5% CAGR |
|
Share in parent market |
22% |
The Orally Disintegrating Tablet Market is likely to have a CAGR of 8.5% over the forecast period. One of the primary factors driving the growth of Orally Disintegrating Tablets is that these tablets are increasingly being used as an alternative to traditional tablets or capsules. Apart from this, the rising adoption of technologically improved products and an increase in the prevalence of CNS illnesses such as depression, migraine, Parkinson's disease, Alzheimer's disease, and schizophrenia are likely to drive market growth.
“Increased Adoption of Orally Disintegrating Tablet ”
Because of improved patient compliance, orally disintegrating tablets (ODTs) are being increasingly adopted over the last few years as a preferable substitute to traditional tablets and capsules. Orally disintegrating tablets dissolve in the mouth within minutes and do not require water to ingest. Traditional medications are difficult to swallow for infants, young children, and elderly people. To address such complications, various orally disintegrating pills with increased performance are developed and launched. Furthermore, advancements like enhanced taste and disintegration are enhancing ODT adoption.
“Enhanced Drug Bioavailability”
The concentration of the medicines taken orally is substantially lowered medication before it reaches the bloodstream and this is where the capacity of Orally Disintegrating Tablets to avoid first-pass metabolism helps solve such difficulties. Orally disintegrating tablets with enhanced bioavailability for protein and peptide-based therapies are a viable alternative for delivering high molecular weight medicines and this factor is likely to drive the growth of the Orally Disintegrating Tablet Market in the coming years.
“Rise in Clinical Trials”
Over the last few years, the popularity of ODTs has increased significantly. The growing number of clinical trials and clinical research are increasing the investment in developing better quality orally disintegrating tablets, and it is likely to improve market growth in the near future.
The Orally Disintegrating Tablet market is highly fragmented and consists of several major and new market players. The market players are focusing on discovering NCEs and excipients for rare diseases along with developing novel formulations to extend the patent regime.
| Attribute | Details |
|---|---|
|
Forecast Period |
2022-2025 |
|
Historical Data Available for |
2012-2021 |
|
Market Analysis |
US$ Mn/Bn for Value |
|
Key Regions Covered |
|
|
Key Countries Covered |
|
|
Key Market Segments Covered |
By Distribution Channel |
|
Key Companies Profiled |
|
|
Customization & Pricing |
Available upon Request |
Orally Disintegrating Tablet Market By Drug Class
Orally Disintegrating Tablet Market By Disease Indication
Orally Disintegrating Tablet Market By Distribution Channel
Orally Disintegrating Tablet Market By Region
In 2022, the Orally Disintegrating Tablet Market stands at USD 19,627.4 million, and it is expected to reach USD 27,222.9 million by 2025 at a CAGR of 8.5%.
Orally Disintegrating Tablet Market exhibited a 11.4% CAGR between 2012 and 2021.
Increased adoption of orally disintegrating tablets is the key factor driving the growth of the market.
Teva Pharmaceutical Industries Ltd., Novartis AG, AstraZeneca, Mylan N.V., and Pfizer Inc., are among the top players in the market.